Analysis of Results
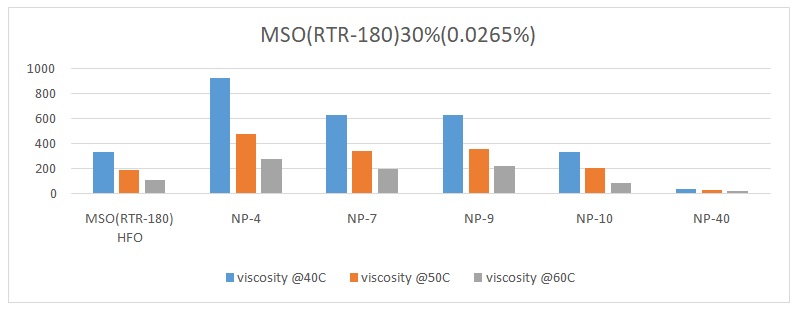
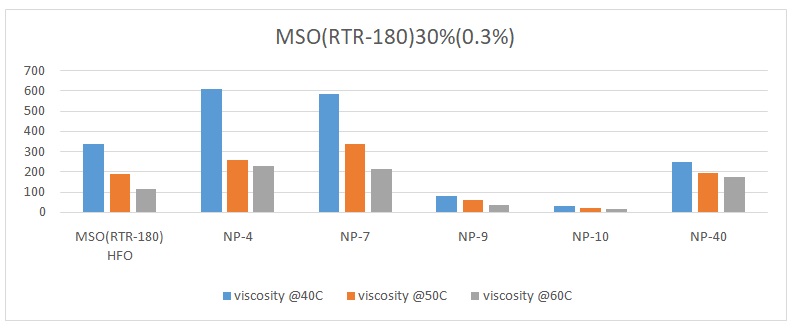
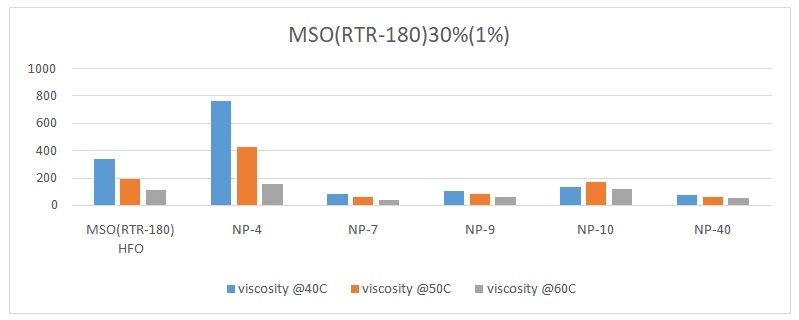
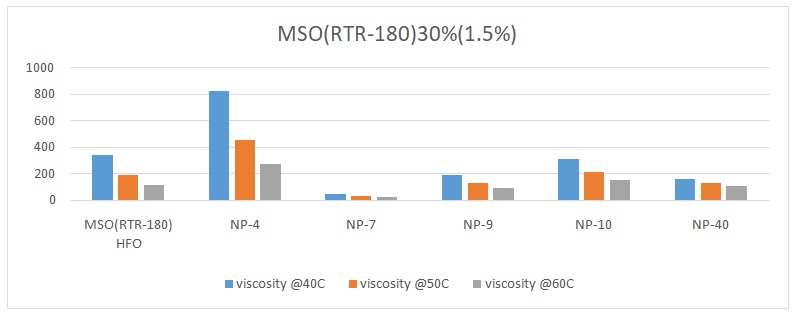
The overall trend was that the viscosity of the oil reduced with an increase in temperature. The viscosity of pure heavy fuel oil (HFO) reduced from 339.25 to 188.7 and finally to 113.38 with an increase in temperature from 40 oC, 50 oC, to 60 oC respectively (Figures 1 to 6). Different surfactants performed differently at various concentrations. For example, NP-40 was more effective at concentrations of 0.0265%, 0.125%, and 1%. On the other hand, NP-7 and NP-9 were highly effective at concentrations of 0.5% and 1%. The lowest viscosity was observed when 0.3% of surfactant NP-10 was added to HFO (RTR-180). At this point, the viscosity readings were 31.1 at 40 oC, 21.58 at 50 oC, and 16.4 at 60 oC (Figure 3). Therefore, it was evident that surfactant NP-10 was the most effective in reducing the viscosity of HFO at 0.3%. Overall, the highest reduction in viscosity was observed in water containing 0.3% surfactant, whereas 0.0125% surfactant yielded high viscosities (Figure 2).


Discussion
The goal of the experiment was to reduce the viscosity of water-oil emulsion consisting of 70% HFO and 30% water using different surfactants at varying concentrations. HFO, which is a residual product, is relatively cheap and undesirable in the fuel industry because of the difficulties associated with its transportation and subsequent processing (HFO, 2017). The chemical composition of HFO, such as large quantities of asphaltenes, leads to a substantial increase in viscosity, which hampers transportation (Santos, Oliveira, & Mansur, 2017). Emulsification combines water and oil in the presence of a surfactant to ease the transportation of oil through pipelines. For the process to be economically viable, the resultant oil in water (O/W) emulsions should contain high oil percentages. Surfactants emulsify the mixture by steadying the droplets of the dispersed phase within the unbroken phase to prevent them from fusing. As a result, stable O/W suspensions with a minimal tension between the interfaces are formed. Surfactants aid in the stabilization of crude oil in the form of microdroplets in the continuous aqueous phase, which lowers the viscosity. Suspensions of crude oil have a “false” plastic or shear-thinning performance, implying that the viscosity is inversely proportional to the shear rate (Santos et al., 2017).
According to Santos et al. (2017), factors that affect crude oil emulsions include temperature, sodium carbonate quantities, the amount of sodium chloride, the proportion of oil, and the total homogenizer revolutions. It was reported that emulsification in water lowered the viscosity of crude oil by 90 to 99% at temperatures of 25 °C, 70% oil and mixing revolution of 22,500 (Santos et al., 2017). However, in this experiment, the viscosity of HFO was lowered by 90.8% in 0.3% surfactant concentration (NP-10) at 70% oil concentration and mixing at 18,000 rpm. The fact that optimal viscosity was attained at a surfactant concentration of 0.3% aligns with the observation by Li, Huang, Ma, and Ji (2016) that maximum surfactant concentration should be kept at 3wt% and oil concentration at 60%. Consequently, increasing the concentration of oil to 70% implied that an effective reduction in viscosity was likely to be attained at lower surfactant concentrations, which was the case in this experiment.
Studies show that the viscosity and stability of a heavy crude oil emulsion are a function of the concentrations of oil and water, temperature, distribution of droplet sizes, and the concentration of the surfactant (Azodi & Nazar, 2013). In this experiment, the oil-water concentration was constant. However, the temperatures and surfactant concentration were varied.
Previous studies have reported that increasing the concentration of surfactants leads to corresponding reductions in viscosity (Santos et al., 2017). The findings of this study corroborated this supposition to some extent. For example, it was noted that increasing the concentration of surfactant NP-10 from 0.0265% to 0.3% led to a significant decrease in viscosity.
The stability and viscosity of water in oil emulsions are a product of the interfacial film surrounding water droplets and the size of the droplet, which are attributed to emulsifiers or stabilizing agents (Li et al., 2016). The characteristics of the interface films are influenced by temperature, pH of water, the compression of the adsorbed film, as well as the constituents of the crude oil. Exposing crude oil to high temperature alters some of its properties, for instance, its stickiness and interfacial tension. Elevated interfacial tension disrupts the integrity of the droplets at the continuous phase, which results in increased movement and merging. Consequently, the viscosity of crude oil reduces. These occurrences explain the observation that the viscosity of the crude oil with and without surfactants increased with an increase in temperature.
The emulsification of heavy crude oil occurs when the oil mixes adequately with water in the presence of an emulsifying agent. Therefore, effective emulsification requires thorough mixing to reduce the sizes of droplets of water, thereby reducing the viscosity of the emulsion (Ashrafizadeh, Motaee, & Hoshyargar, 2012). Consequently, keeping other factors (such as temperature, type and amount of emulsifier, and oil-water ratio), the mixing process plays an important role in the entire process because it affects the type of emulsion formed. In this experiment, however, all the samples were treated equally by mixing at 18,000 rpm for 15 minutes. Therefore, mixing did not influence the viscosity values that were recorded. It has been noted that emulsification techniques such as ultrasonic waves were more effective than mechanical homogenization because of the capacity to generate relatively small dispersed phase droplets (Hoshyargar, Marjani, Fadaei, & Shirazian, 2015). The experiment used mechanical homogenization to emulsify the oil-water mixtures. There is a possibility that using ultrasonic techniques would produce more stable emulsions.
The viscosity of the HFO varied at different surfactant types and concentrations thereby supporting the hypothesis that certain traits of the emulsifying agent such as its nature and quantity have a direct effect on the viscosity of an emulsion. It was noted that the viscosity of the HFO was lowest at the highest temperature. This observation supports the efforts by certain players in the fuel industry to heat pipelines to reduce the viscosity of the heavy crude oil. However, the heating of pipelines is not cost effective as it uses up extra energy in the heating process. Additionally, heating has been reported to lower the stability of emulsion created (Santos et al., 2017). Therefore, surfactants that achieve low viscosities at low temperatures are the best option. According to Muñoz, Ancheyta, and Castañeda (2016), crude oil should have a maximum viscosity ranging from 250 and 400 cSt at 37.8 °C for smooth pipeline transportation. HFO (RTR-180) has a maximum viscosity 700 cSt at room temperature (HFO, 2017). Based on this estimation, it can be concluded that the surfactant NP-40 resulted in satisfactory viscosities of the HFO at all concentrations and temperatures.
References
Ashrafizadeh, S., Motaee, E., & Hoshyargar, V. (2012). Emulsification of heavy crude oil in water by natural surfactants. Journal of Petroleum Science and Engineering, 86(87), 137-143.
Azodi, M., & Nazar, A. R. S. (2013). An experimental study on factors affecting the heavy crude oil in water emulsions viscosity. Journal of Petroleum Science and Engineering, 106, 1-8. Web.
HFO. (2017). Web.
Hoshyargar, V., Marjani, A., Fadaei, F. & Shirazian, S. (2015). Prediction of flow behavior of crude oil-in-water emulsion through the pipe by using rheological properties. Oriental Journal of Chemistry, 28(1), 109-113.
Li, C., Huang, Q., Ma, S., & Ji, C. (2016). An experimental study on the viscosity of water-in-oil emulsions. Journal of Dispersion Science and Technology, 37(3), 305-316.
Muñoz, J. A., Ancheyta, J., & Castañeda, L. C. (2016). Required viscosity values to ensure proper transportation of crude oil by pipeline. Energy & Fuels, 30(11), 8850-8854.
Santos, I. C. V. M., Oliveira, P. F., & Mansur, C. R. E. (2017). Factors that affect crude oil viscosity and techniques to reduce it: A review. Brazilian Journal of Petroleum and Gas, 11(2), 115-130.
